TEXAS CARES HAS NOW ENDED
Lee está página de web en español.
We are so grateful for our Texas CARES participants. Through their time and effort, we reached a better understanding of how COVID-19 impacted Texas families and communities.
While this phase of Texas CARES is over, the new phase has begun. Texas CARES is now Health of Texas. Health of Texas aims to understand the state of health, chronic disease, and health-related needs for Texans. Visit the Health of Texas website here. Watch a video about Health of Texas here.
WHAT WAS TEXAS CARES?
The Texas Coronavirus Antibody REsponse Survey (CARES) sought to understand the human antibody response to SARS-CoV-2, the virus that causes COVID-19. Since September 2020, more than 90,000 Texans ages 6 months to 90 years old took part. Over multiple time points, survey participants helped us measure things like how many people in the survey had COVID-19 antibodies and how long those antibodies lasted. The timeline below illustrates how Texas CARES worked.
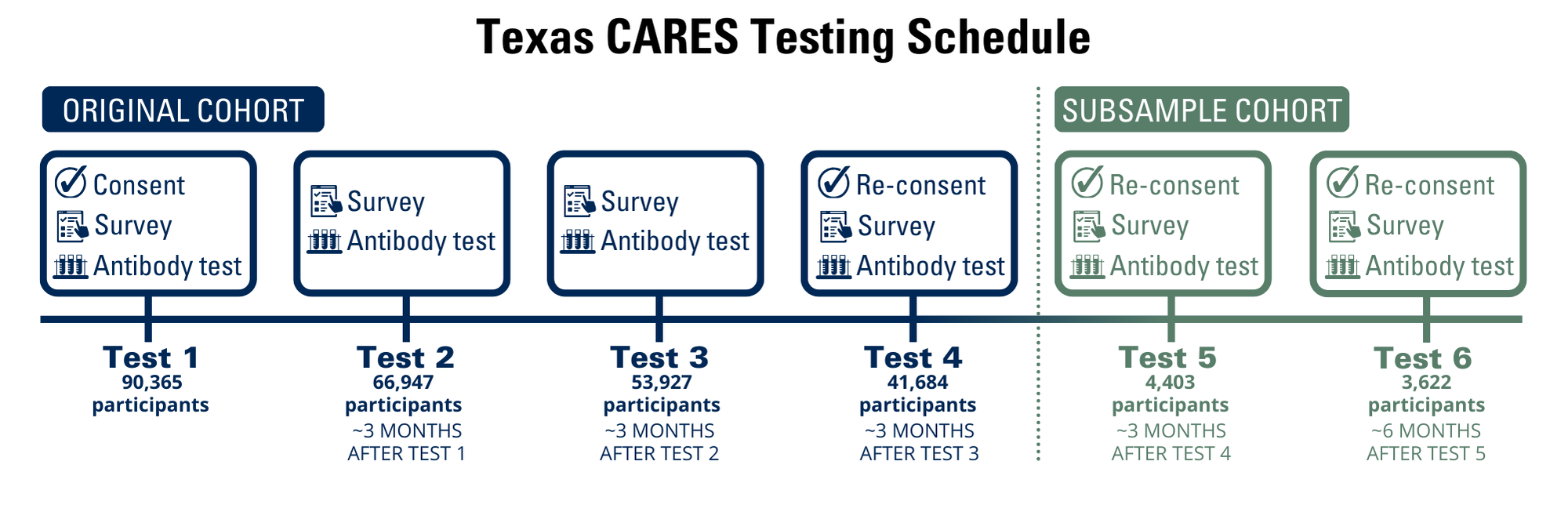
Data collection and analysis
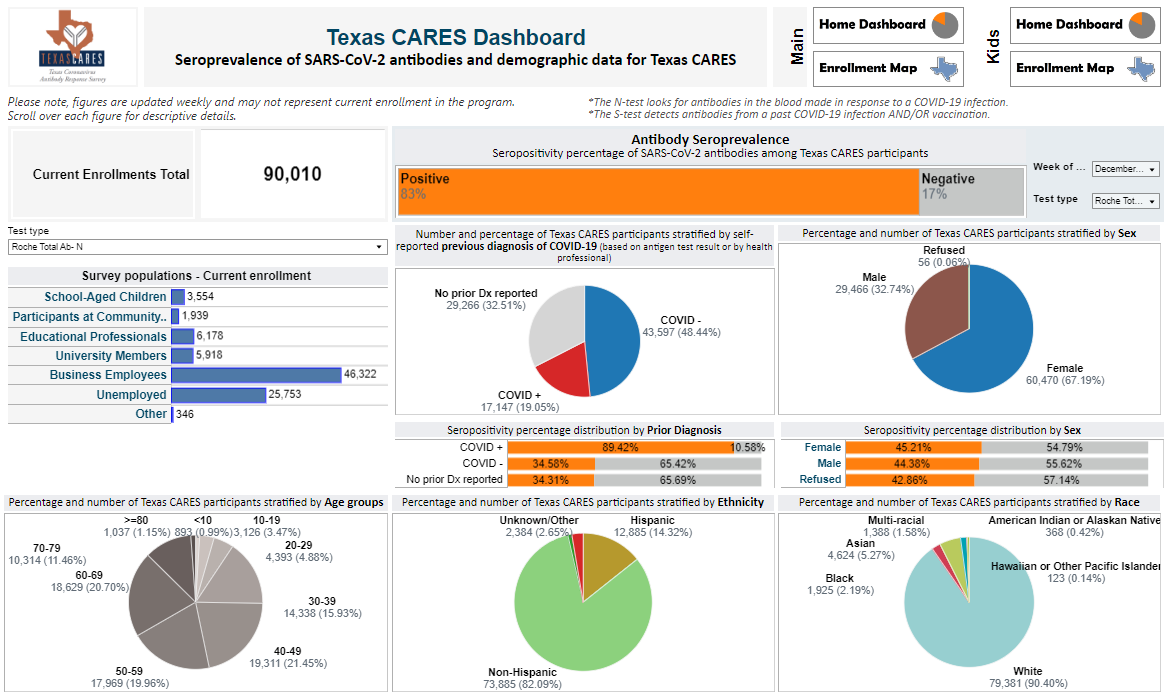
We collected and organized the information we learned from participants in a dashboard accessible to everyone. While this information will no longer be updated, you can still revisit the data over various timepoints — including data for Texas CARES Kids participants — on the dashboard page.
WHAT DID TEXAS CARES LEARN?
Thanks to our participants, we learned so much about COVID-19 over the course of Texas CARES. We identified better ways to estimate COVID-19 infections in communities. We analyzed the most common COVID-19 variant symptoms in children. And we observed the prozone effect — very high (strongly positive) antibody levels that test as weakly positive — on some S-test results, resulting in the use of a new testing process.
Look back on the news Texas CARES made over each of the four years of the survey:
Read a complete list of our findings here. Click on each title in the PDF to read the article that was published.
Check out our collection of Texas CARES resources here, including coloring sheets, Long COVID information, and more.
Most of all, we learned that Texans know how to come together when faced with a challenge. The participants who made up Texas CARES gave freely of their time and effort to contribute to science when we were all faced with a new pandemic. They were part of something big, and their contributions were key in understanding how COVID-19 affected people and communities throughout the state.
We look forward to continuing to connect with participants during the next phase: Health of Texas!

.png?language_id=1)
