Hypoplastic Left Heart Syndrome (HLHS)
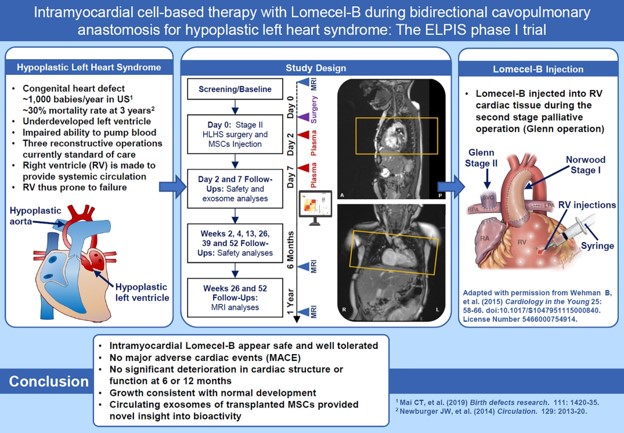
One of the most severe forms of congenital heart disease is Hypoplastic Left Heart Syndrome (HLHS). Congenital diseases are those which are present at birth. Approximately 1000 babies are born each year with HLHS (this is about 1 out of every 3,841 babies born in the US each year). Babies with HLHS have hearts that are severely underdeveloped on the left side; resulting in the right side of the heart compensating to get blood to the lungs and the rest of the body. Infants born with HLHS undergo a series of staged operations during the first few years of life aimed at reconstructing the heart to improve circulation.
For further information: https://www.cdc.gov/ncbddd/heartdefects/hlhs.html


The ELPIS II Trial
The study is funded by the National Heart, Lung, and Blood Institute (part of the National Institutes of Health). The purpose of this study is to test whether Lomecel-B works in treating patients with HLHS and to gather additional information about the safety of Lomecel-B. Provided by the clinical-stage biotechnology company Longeveron Inc, Lomecel-B contains human mesenchymal stem cells (MSCs) as the active ingredient. MSCs are special cells in the body that are able to change into other types of cells, such as heart, blood, and muscle cells. MSCs are found in various tissues of the body, such as the bone marrow, which is the spongy tissue inside of your bones. Lomecel-B uses MSCs from bone marrow of unrelated young healthy donors. These are called “allogeneic”, and do not require donor matching to the patient. Lomecel-B is injected into the heart during the child’s second scheduled HLHS surgery (at approximately 4-6 months of age). The goal of the research is to demonstrate whether Lomecel-B improves heart function more than the standard surgical treatment.
Participating in the ELPIS II trial
Thirty-eight children with HLHS are expected to participate in this study at seven research sites across the United States. Participants will come to the center for two brief visits during weeks leading up to the Phase II palliation operation to review their health information and collect information needed to determine safety of participating. The child will be randomized (like flipping a coin) to receive Lomecel-B during their operation or to undergo their operation without receiving cells. It is not known whether the cells will be of benefit. For this reason, some study participants may not receive the cells. This will allow a careful comparison to study the benefits and side effects of the Lomecel-B. Follow-up visits will take place at four timepoints over the 12 months following this surgery.

Where can I learn more about participating?
Clinical centers participating in the ELPIS II trial are shown below. Contact us to learn more about the trial.
Ann & Robert H. Lurie Children’s Hospital of Chicago
Carmen Diaz-Leos
[email protected]
Primary Children's Hospital/University of Utah
Linda Lambert
[email protected]
Children's Hospital Los Angeles
Carly Weaver
[email protected]
Advocate Children’s Hospital
Bonnie Hughes
[email protected]
Boston Children’s Hospital
Michael Bamgbose
[email protected]
Children's Healthcare of Atlanta
Emily Klingman
[email protected]
UTHealth McGovern Medical School
Rebecca Sam
[email protected]
Ann & Robert H. Lurie Children’s Hospital of Chicago - Clinical Coordinating Center
Stephanie Brazis
[email protected]
UTHealth School of Public Health - Data Coordinating Center
Kiran Mansoor
[email protected]