About participation in Texas CARES
-
Can I still join Texas CARES?
We have reached enrollment goals for this portion of the program and have ended new enrollment. If you have already joined, you can complete your remaining surveys and tests. You can check for information about new enrollment on our website, or stay subscribed to Texas CARES texts and emails for updates.
-
Who can join Texas CARES?
Texans between the ages of 6 months and 90 years old can join, even if they have never been tested for COVID-19 or have tested positive or negative for COVID-19 in the past. You can join even if you have been vaccinated or plan to get the vaccine or a booster soon.
-
Why should I continue to take part in Texas CARES?
Being a part of Texas CARES will help Texans understand how many people may have had COVID-19 and did not show symptoms, how many Texans have COVID-19 antibodies (either from infection or the vaccine), and how long the antibodies will last. It may also help us to better understand long COVID, which some people have.
-
Can I join if I have gotten or plan to get a COVID-19 vaccine booster?
Yes! For the most accurate antibody test results, it is best to wait at least 14 days after your booster to get your antibody test.
-
Can I be a part of Texas CARES if I am already in another study?
Yes, you can still join Texas CARES if you are already in another study.
-
How long should I wait to get my antibody test at CPL after I test positive for COVID-19?
We suggest waiting at least 14 days after a positive COVID-19 viral test before getting your antibody test. Antibodies may not be present in those tested early in the infection and may result in a negative N-test.
-
How do I contact the Texas CARES team?
You can contact the Texas CARES team by email at [email protected] or by leaving a voicemail on our secure line at 713-500-9441. We will reach out to you as soon as possible.
How Texas CARES works
-
Why are there multiple antibody tests?
Texas CARES aims to identify possible protection from future COVID-19 infection given by antibodies that your body makes in response to a COVID-19 infection or vaccine. Texas CARES looks at how long those antibodies stay in your body over time with a survey and antibody test every three months.
-
How will I know when to get my next antibody test?
Texas CARES is designed to test participants at multiple time points, about three months apart. Here is how it works:
- You will get a text reminder with your link to the next survey three months after your last results are received.
- Once you fill out your new survey, you will get a text with your new order number to take to CPL for your next test.
We ask that you take your test as close to the three-month mark as possible. Please email us at [email protected] and include your cellphone number and date of birth if you have not received your text message three months after your last antibody test.
Note: At this time, most participants have done four surveys and antibody tests. Some participants were selected to do more surveys and antibody tests. Stay subscribed to Texas CARES emails and texts for future updates.
-
I filled out a survey a while ago, but I haven’t gone to the lab yet. Can I still go for my first antibody test?
We have closed past testing windows for the previous surveys and antibody tests, and unused orders have expired. Please check our website and social media for updates on the program, data collected, and future enrollment.
-
I have gotten four antibody tests. Will I get another survey and antibody test?
Some participants were selected to complete fifth and sixth surveys and antibody tests, about six months apart. Please check our website or social media for future updates, and stay subscribed to Texas CARES emails and texts for more information.
-
How do I redo my survey if anything changes?
If you did your survey more than 30 days ago and have not gotten your antibody test, or need to make changes to your survey due to a recent COVID-19 infection or new vaccine or booster, please email Texas CARES at [email protected] to receive a new survey link.
-
Do I need to pay for my test at CPL?
No, expenses are covered by Texas CARES.
-
Do I need to make an appointment for my antibody test?
No, but we suggest checking the hours for the CPL locations near you by clicking here before going to the lab. No appointment is needed.
-
What do I need to bring to CPL?
Bring your picture ID and your text message that contains your test ID# 255799-XXXXXXXX.
-
How will I get my results?
Your results (positive or negative for COVID-19 antibodies) will be texted to you 2-5 days after you get your blood drawn at CPL. You can view the full report on CPL’s portal after you receive the text message from Texas CARES. If your results are not loaded in the portal, please call the Sonic MyAccess customer service group at 844-280-8484.
Parents or guardians may create an account for their?minor child, aged 17 or younger, by clicking here. You will be asked to complete, sign, and submit a?Record Request (RR) form along with proper identification via email, fax, or mail. For assistance with this process, please call 844-280-8484.
About your test results
-
What does a negative antibody test result mean?
N-test: A negative N-test means you may not have had COVID-19 before. In some people, antibody levels may decrease and not be detected over time. The test may not detect antibodies early in an infection.
S-test: A negative S-test means the test did not detect antibodies from either COVID-19 infection or the vaccine.
Read more from the CDC about antibody tests by clicking here.
-
What does a positive antibody test result mean?
N-test: A positive N-test means you may have had a previous COVID-19 infection.
If you did not have COVID-19 symptoms but tested positive for antibodies, you may have had an infection in the past.
S-test: A positive S-test means you have antibodies from a past COVID-19 infection or vaccine.
Read more from the CDC about antibody tests by clicking here.
-
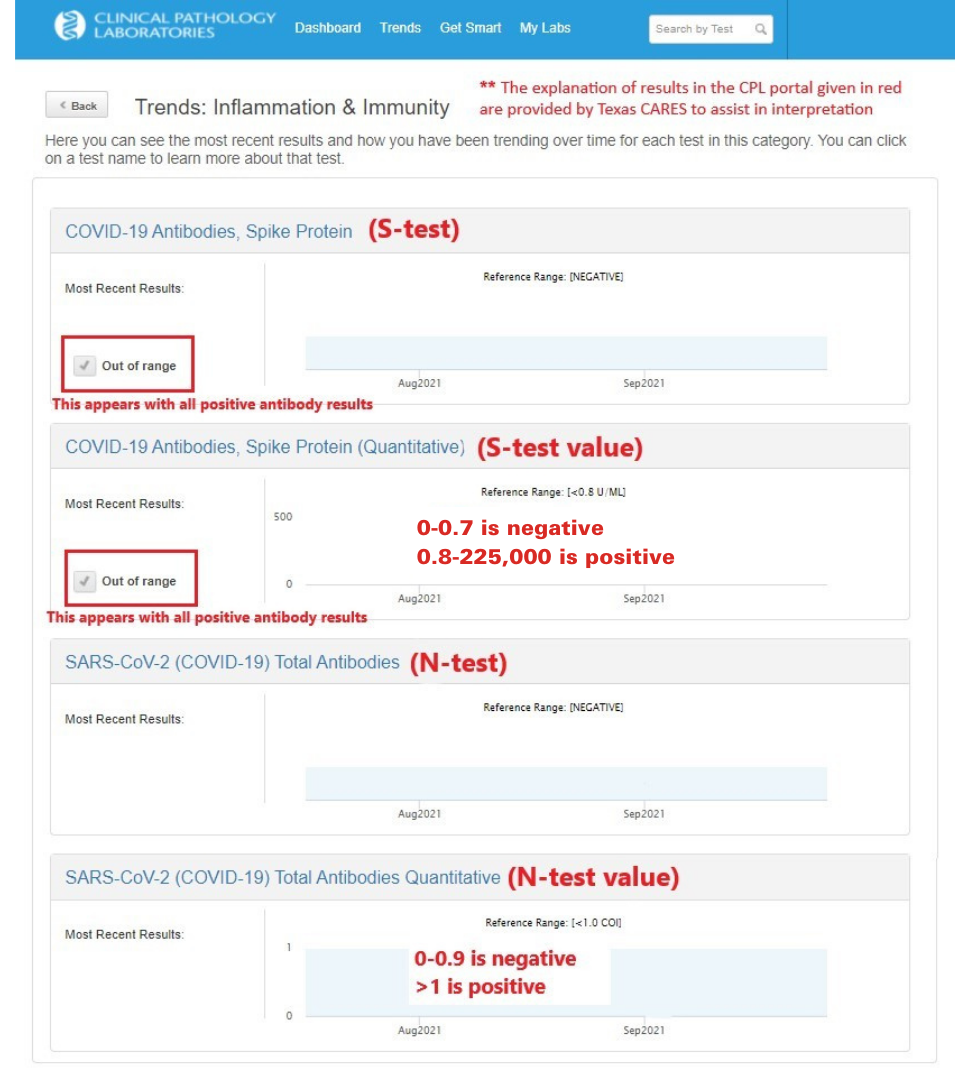
What do my CPL test results mean?
"COVID-19 Antibodies, Spike Protein" (S-Test): detects antibodies from a past COVID-19 infection AND/OR vaccination
"SARS-CoV-2 Total Antibodies" (N-Test): detects ONLY antibodies from a past COVID-19 infection
The sample below may help interpret your results in the portal.
CPL has recently increased the S-test range. The S-test measures antibodies made after a COVID-19 infection and/or vaccine.
You may see the message below from CPL in your results:
EXPANDED CLINICAL REPORTING RANGE OF 0.4-225,000 IU/ML
HAS BEEN VALIDATED TO REPLACE PRIOR REPORTING RANGE OF
0.4-25,000 IU/ML, EFFECTIVE 8/8/2023.
S-antibody levels increased during the pandemic. Your previous tests listed your S-test value as 0.8-25,000 IU/mL. The new range will measure values up to 225,000 IU/mL.
This new process for testing S-antibodies will give you an improved picture of your current antibody level. It will not affect previous results.
We are unable to give medical advice. Please follow up with your primary care provider if you have questions about your results.
-
What does the S-test number mean?
The S-test shows the number of antibodies in your system at the time of the test made after either your COVID-19 vaccine or past infection. There is no guidance on which levels are protective. We are not able to give medical advice. Please follow up with your care provider if you have questions about your results.
You can use the S-test to:
- Confirm the presence of antibodies.
- Track your levels over time.
- Help Texans understand and identify the level of antibodies needed to protect from severe infection.
-
What does the "SARS-CoV-2 Total Antibodies Quantitative" (N-test) COI number mean?
The N-test measures and determines if antibodies (IgM, IgG, IgA) to the COVID-19 N-protein are present in the sample. The N-test cutoff index (COI) helps to identify a positive or negative sample. Positive results mean you may have had COVID-19 before. Antibodies from the COVID-19 vaccine will not show up on this test.
COI* < 1.0 = non-reactive (or negative)
COI ≥ 1.0 = reactive (or positive)
Numeric result Result message Interpretation
COI < 1.0 Non-reactive Negative for anti-SARS-CoV-2 antibodies COI ≥ 1.0 Reactive Positive for anti-SARS-CoV-2 antibodies Information from Roche (the test manufacturer)
Interpretation of the results:
Results obtained with the Elecsys Anti-SARS-CoV-2 assay can be interpreted as follows:
The magnitude of the measured result above the cutoff is not indicative of the total amount of antibody present in the sample. The individual immune response following SARS-CoV-2 infection varies considerably and might give different results with assays from different manufacturers. Results of assays from different manufacturers should not be used interchangeably.
The COI number should not be used to judge or estimate the amount of antibodies present in the blood draw. The COI number is used to identify a positive or negative sample.
-
What COVID-19 antibody test is used?
- S-test: Roche Elecsys Anti-SARS-CoV-2 S (semi-quantitative assay that detects antibodies from vaccination or past infection).
- N-test: Roche Elecsys Anti-SARS-CoV-2 (qualitative assay that only detects antibodies from past infection).
More information about these tests:
-
If I got the vaccine, does a negative antibody result mean the vaccine did not work?
No. It usually takes about two weeks after a vaccine to get a full antibody response, and this timeline varies between individuals.
-
What is the accuracy of the COVID-19 antibody test?
Analytical Specificity: 99.5%
Clinical Sensitivity at least 14 days after confirmed test: 99.5%
S-test: (infection and vaccine)
Analytical Specificity: 100%
Negative Percent Agreement: 99.9%
Positive Percent Agreement: 96.6%
-
Will my results be shared with anyone?
Your personal results will not be shared with your employer or school.
General COVID-19 information
-
What is the difference between SARS-CoV-2 and COVID-19?
SARS-CoV-2 is the virus that causes COVID-19.
-
What is an antibody?
An antibody is a protein made by your body in response to a specific foreign substance, such as a virus, that helps your body fight infection. Antibodies in the blood can help identify a past infection. Antibodies are also made in response to vaccines.
-
What is the difference between an antibody test and a viral test?
Antibody tests show signs of a past infection and/or a vaccine. Viral tests detect an active infection.
-
Where can I go for updates and more information about COVID-19?
The latest information on COVID-19 is available from the CDC, which you can visit by clicking here.

