About the Coordinating Center for Clinical Trials
Welcome
The Coordinating Center for Clinical Trials (CCCT) provides full-service trial coordination for research studies. We handle the logistical and statistical challenges of coordinating small and large multicenter studies and research projects, allowing Investigators to focus on their patient-participants. Our expert team collaborates with investigators every step of the way – from study design and execution to the dissemination of trial findings. We invite you to learn more about our services, our team, and who we can expand your research, streamline your data collection, and manage the operations integral to your project.
Who we are
The UTHealth Coordinating Center for Clinical Trials (CCCT) has an established history of advancing science through coordinating clinical trials. Established in 1971, the CCCT has served as a full-service coordinating center for numerous nationwide multicenter clinical trials, both large and small. Our expertise spans research areas including biostatistics, epidemiology, clinical trials, data management, trial design, data science, computer science, bioinformatics, and genomics. The mission of the CCCT is to improve a broad spectrum of public health interests through the coordination of clinical trials, collaboration with clinical investigators, and the development of statistical and trial methodology.
Clinical trials are undergoing a revolution in the era of big data. Here at the CCCT, we have the capacity to undertake multifaceted clinical trials using complex and high-dimensional data gathered from sources including wearable devices and electronic medical records. Leveraging data that’s collected automatically or as part of the patient standard of care, allows for clinical trials that are more efficient and less costly to design.
Research interests
The CCCT has the capability and expertise to work on many topics of medical and public health interests.
Our focus is in leading and conducting multicenter clinical trials and research studies. We provide academic expertise and leadership coupled with full-service clinical trial management capabilities, data and adverse event monitoring, and statistical analysis support for publications.
Our ultimate goal is in developing and sharing knowledge to improve patient care and public health. We accomplish this through the seamless integration of disease area expertise, innovative interventions, and sound clinical trial methodology.
Areas of expertise
- Cardiovascular
- Diabetes
- Kidney disease
- Stroke
- Sickle cell
- Ophthalmology
- Cell Therapy
- Trauma
- Rheumatology
- Neurology
- Dental
- Psychiatry
- Gastroenterology
- Pulmonary
- Pharmacokinetics
Innovative interventions
- Diet-drug trials
- Community-based clinics
- Dissemination of trial results
- Wearable device trials
- Smartphone-based intervention to reduce recidivism
- Strategies for recruitment of racial and ethnic diverse groups in clinical trials
- Randomized trial for an intervention to decrease sedentary behavior
Clinical trial methodology
- Factorial design
- Bayesian adaptive design
- Adaptive randomization, seamless trial
- Group sequential monitoring
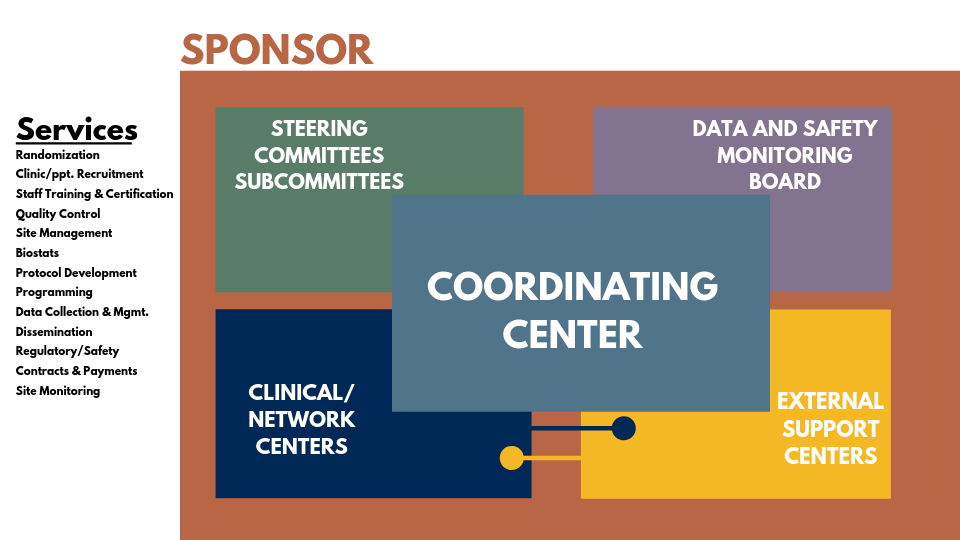
Services offered by the CCCT
50 years of successfully providing comprehensive services and coordinating large multi-center clinical trials
Although our primary goal is to design and assist with large multicenter clinical trials, we also work with smaller and single-site trials. Regardless of trial size and complexity, we provide high-quality study design, monitoring, management, reporting, and interpretation of study findings.
Our model (below) highlights our capabilities and can be scaled to fit the needs of a given research project. Take a look at what we can do for you.
Consulting
First hour: free of charge
Collaborations
Free for collaboration on the grant application, but the service provider and his/her associates’ effort and other needs for the proposed project should be appropriately budgeted in the grant application. We are happy to arrange a meeting with you to discuss the project and provide a quote for services that meet the needs of the trial.
Trials and projects
Antihypertensive and Lipid Lowering Treatment to Prevent Heart Attack Trial (ALLHAT)
Read about ALLHAT here.
Ancillary Study. Long-term Benefits & Harms of Antihypertensive Drugs in the Elderly: Up to 22-year Follow-Up of ALLHAT Trial Participants. Xainglin Du, PI. NIA. 2018-2022 ($1,324,000).
Evaluation of Lomecel-B™ Injection in Patients with Hypoplastic Left Heart Syndrome: A Phase IIb Clinical Trial (ELPIS II)
The ELPIS II trial is being conducted by a consortium of clinical centers in the US who routinely direct surgical and follow up care for children with hypoplastic left heart syndrome (HLHS). The trial is testing whether Lomecel-B (containing human mesenchymal stem cells) is safe and effective in treating patients with HLHS when administered during the child’s second staged palliative surgery.
Sponsored by the National Heart, Lung and Blood Institute (NHLBI); 2020-2025 ($1,862,493).
A Phase IIB Randomized, Placebo-Controlled, Multicenter Study of the Comparative Efficacy and Safety of Transendocardial Injection of Allogeneic-MSC versus Placebo in Patients with Non-Ischemic Dilated Cardiomyopathy (DCM II)
The purpose of the DCM II study is to determine whether giving allogeneic mesenchymal stem cells to patients with heart muscle damage is safe and whether this improves heart function. Additionally, this study will examine whether an individual’s genetics plays a role in determining his/her response to the cell therapy. Four US clinical centers are participating.
Sponsored by the US Department of Defense; 2020-2024 ($4,778,129).
Autologous Cardiac Stem Cell Injection in Patients with Hypoplastic Left Heart Syndrome (CHILD)
The CHILD study is assessing the safety, feasibility, and efficacy of intramyocardial delivery of c-kit+ cells in patients with hypoplastic left heart syndrome under 6 months of age.
Sponsored by the Marcus Foundation Inc.; 2019-2023 ($761,772).
The Benefits of Stroke Treatment Delivered by a Mobile Stroke Unit Compared with Standard Management by Emergency Medical Services (BEST-MSU) Trial
This is an observational, prospective, multicenter, alternating-week trial comparing Mobile Stroke Units with Emergency Medical Services management. The study enrolled 1,515 patients, of which the primary analysis focused on the 1,047 that were tPA-eligible. The overall goal of the study is to determine whether Mobile Stroke Units lead to lower disability after 90 days of stroke as well as fewer healthcare utilizations, and whether they are cost-effective.
Sponsored by Patient-Centered Outcomes Research Institute (PCORI), 2015-2022 ($7,997,773).
CATCH Healthy Smiles: An Elementary School Oral Health Intervention Trial
This is a cluster-randomized trial conducted in 26 schools. The purpose of this study is to clinically evaluate the effects of a school-based behavioral intervention, CATCH Healthy Smiles, to reduce the risk of dental caries in a cohort of kindergarten through 2nd grade (K-2) children serving low-income, ethnically-diverse children, to determine the impact of CATCH Healthy Smiles on child behavioral, psychosocial, and environmental outcomes beginning in K through 2nd grade, compared to children in the control schools and to examine the extent to which the child behavioral, psychosocial, and environmental factors mediate the improvements in child caries risk.
Sponsored by National Institute of Dental and Craniofacial Research (NIDCR), 2019-2025 ($2,257,769)
Trauma Resuscitation with Low-Titer Group O Whole Blood or Products (TROOP Trial)
TROOP is a pragmatic, multicenter, Bayesian, group-sequential, combined non-inferiority/ superiority, randomized clinical trial. The trial will enroll approximately 1100 injured patients with hemorrhagic shock anticipated to require massive blood transfusions. The overall goal of the study is to determine whether patients who are critically injured and need a large transfusion of blood should be given whole blood, or blood that has been separated into its individual components (red cells, platelets, plasma, etc.). Participants will be randomized to receive either whole blood (at least 8 units) or only component therapy and will be followed for 30 days to assess outcomes.
Sponsored by National Heart, Lung, and Blood Institute (NHLBI), 2022-2027 ($653,807 in Y1).
Gap-Based Milieu Biomarkers for Traumatic Brain Injury (GAMBIT-TBI)
GAMBIT-TBI is a multi-center and multi-project diagnostic clinical study.?The goal of GAMBIT-TBI is to discover new blood- and imaging-based biomarkers to identify traumatic brain injury subphenotypes based on their recovery trajectories. Sponsored by Department of Defense US Army Medical Research Acquisition Activity, 2022-2026 ($865,872).
Recent publications and collaborations
-
DeSantis Stacia
2024
- #Patel, R. R., Delclos, G. L., DeSantis, S. M., Cannell, M. B., Lupo, P. J., Bishop, A. J., Lazar, A. J., Lin, P. P., Benjamin, R. S., Patel, S. R., Ludwig, J., Ravi, V., Livingston, J. A., Somaiah, N., Zarzour, M. A., Conley, A. P., & Araujo, D. M. (2024). Does the primary tumor site drive biology for patients with synovial sarcoma? American Journal of Clinical Oncology. Advance online publication. https://doi.org/10.1097/COC.0000000000001142
- Messiah, S. E., Abbas, R., Bergqvist, E., Kohl, H. W. III, Swartz, M. D., Talebi, Y., Sabharwal, R., Han, H., Valerio-Shewmaker, M. A., DeSantis, S. M., Yaseen, A., Gandhi, H. A., Amavisca, X. F., Ross, J. A., Padilla, L. N., Gonzalez, M. O., Wu, L., Silberman, M. A., Lakey, D., Shuford, J. A., Pont, S. J., & Boerwinkle, E. (2024). Factors associated with elevated SARS-CoV-2 immune response in children and adolescents. Frontiers in Pediatrics, 12, 1393321. https://doi.org/10.3389/fped.2024.1393321
- Messiah, S. E., Talebi, Y., Swartz, M. D., Sabharwal, R., Han, H., Bergqvist, E., Kohl, H. W. III, Valerio-Shewmaker, M., DeSantis, S. M., Yaseen, A., Kelder, S. H., Ross, J., Padilla, L. N., Gonzalez, M. O., Wu, L., Lakey, D., Shuford, J. A., Pont, S. J., & Boerwinkle, E. (2024). Long-term immune response to SARS-CoV-2 infection and vaccination in children and adolescents. Pediatric Research, 96(2), 525–534. https://doi.org/10.1038/s41390-023-02857-y
- #Hahn, G., Prokopenko, D., Hecker, J., Lutz, S. M., Mullin, K., Sejour, L., Hide, W., Vlachos, I., DeSantis, S. M., Tanzi, R. E., & Lange, C. (2024). Prediction of disease-free survival for precision medicine using cooperative learning on multi-omic data. Briefings in Bioinformatics. (Accepted for publication).
- Hahn, G., Prokopenko, D., Hecker, J., Lutz, S. M., Mullin, K., Tanzi, R. E., DeSantis, S. M., & Lange, C. (2024). Polygenic hazard score models for the prediction of Alzheimer’s free survival using the lasso for Cox’s proportional hazards model. Genetic Epidemiology. Advance online publication.
- Dixon, A., Kenny, J. E., Buzzard, L., et al., & PROPPR Study Group (including DeSantis, S. M.). (2024). Acute respiratory distress syndrome, acute kidney injury, and mortality after trauma are associated with increased circulation of syndecan-1, soluble thrombomodulin, and receptor for advanced glycation end products. Journal of Trauma and Acute Care Surgery, 96(2), 319–325.
- Yaseen, A., DeSantis, S. M., Sabharwal, R., Talebi, Y., Swartz, M. D., Zhang, S., Leon Novelo, L., Pinzon-Gomez, C. L., Messiah, S. E., Valerio-Shewmaker, M., Kohl, H. W. III, Ross, J., Lakey, D., Shuford, J. A., Pont, S. J., & Boerwinkle, E. (2024). Baseline characteristics of SARS-CoV-2 vaccine non-responders in a large population-based sample. PLoS One, 19(5), e0303420. https://doi.org/10.1371/journal.pone.0303420
- #Patel, R. R., Delclos, G. L., DeSantis, S. M., Cannell, M. B., Lupo, P. J., Lin, P. P., & Araujo, D. M. (2024). Epidemiological trends of synovial sarcoma by primary tumor sites in the U.S. from 2000 to 2020. Cancer Epidemiology. (In press).
2023
- DeSantis, S. M., Yaseen, A., Swartz, M. D., et al. (2023). Incidence of SARS-CoV-2 breakthrough infections after vaccination in adults: A population-based survey through March 1, 2023. Open Forum Infectious Diseases. (Accepted for publication).
- DeSantis, S. M., Yaseen, A., Hao, T., León-Novelo, L., Talebi, Y., Valerio-Shewmaker, M. A., Pinzon Gomez, C. L., Messiah, S. E., Kohl, H. W., Kelder, S. H., Ross, J. A., Padilla, L. N., Silberman, M., Tuzo, S., Lakey, D., Shuford, J. A., Pont, S. J., Boerwinkle, E., & Swartz, M. D. (2023). Incidence and predictors of breakthrough and severe breakthrough infections of SARS-CoV-2 after primary series vaccination in adults: A population-based survey of 22,575 participants. Journal of Infectious Diseases, 227(10), 1164–1172. https://doi.org/10.1093/infdis/jiad020
- Messiah, S. E., Talebi, Y., Swartz, M. D., Sabharwal, R., Han, H., Bergqvist, E., Kohl, H. W. III, Valerio-Shewmaker, M., DeSantis, S. M., Yaseen, A., Kelder, S. H., Ross, J., Padilla, L. N., Gonzalez, M. O., Wu, L., Lakey, D., Shuford, J. A., Pont, S. J., & Boerwinkle, E. (2023). Long-term immune response to SARS-CoV-2 infection and vaccination in children and adolescents. Pediatric Research. Advance online publication. https://doi.org/10.1038/s41390-023-02857-y
- #Hebballi, N. B., DeSantis, S., Brown, E. L., Markham, C., & Tsao, K. (2023). Body mass index is associated with pediatric complicated appendicitis and postoperative complications. Annals of Surgery, 278(3), 337–346. https://doi.org/10.1097/SLA.0000000000005965
- Yaseen, A., Robertson, C., Cruz Navarro, J., Chen, J., Heckler, B., DeSantis, S. M., Temkin, N., Barber, J., Foreman, B., Diaz-Arrastia, R., Chesnut, R., Manley, G. T., Wright, D. W., Vassar, M., Ferguson, A. R., Markowitz, A. J., & Yamal, J. M. (2023). Integrating, harmonizing, and curating studies with high-frequency and hourly physiological data: Proof of concept from seven traumatic brain injury data sets. Journal of Neurotrauma. Advance online publication. https://doi.org/10.1089/neu.2023.0023
- #Xie, L., Chandrasekhar, A., DeSantis, S. M., Almandoz, J. P., de la Cruz-Muñoz, N., & Messiah, S. E. (2023). Discontinuation and reduction of asthma medications after metabolic and bariatric surgery: A systematic review and meta-analysis. Obesity Reviews, 24(2), e13527. https://doi.org/10.1111/obr.13527
- #Tadese, B. K., DeSantis, S. M., Mgbere, O., Fujimoto, K., & Darkoh, C. (2022). Clinical outcomes associated with co-infection of carbapenem-resistant Enterobacterales and other multidrug-resistant organisms. Infection Prevention in Practice, 4(4), 100255. https://doi.org/10.1016/j.infpip.2022.100255
- Messiah, S. E., Swartz, M. D., Abbas, R. A., Talebi, Y., Kohl, H. W. III, Valerio-Shewmaker, M., DeSantis, S. M., Yaseen, A., Kelder, S. H., Ross, J. A., Padilla, L. N., Gonzalez, M. O., Wu, L., Lakey, D., Shuford, J. A., Pont, S. J., & Boerwinkle, E. (2023). SARS-CoV-2 serostatus and COVID-19 illness characteristics by variant time period in non-hospitalized children and adolescents. Children, 10(5), 818. https://doi.org/10.3390/children10050818
- Swartz, M. D., DeSantis, S. M., Yaseen, A., et al. (2023). Antibody duration after infection from SARS-CoV-2 in the Texas Coronavirus Antibody Response Survey. Journal of Infectious Diseases, 227(2), 193–201.
^The above paper received 23 citations in less than one year.
-
Tami-Maury Irene
- Afolayan, O. K., Velazquez, J., Tundealao, S., Fernández, E., Martínez, C., Leon-Novelo, L., Retamales, J., Tamí-Maury, I. (2025). Trends and association between smoking and the socio-demographic index among 11 South American countries, 1990-2019. Substance Use & Misuse, 1-11. https://doi.org/10.1080/10826084.2024.2440384
- Tamí-Maury, I., Millett, T. J. (2024). Recommendations and implementation considerations for the routine collection of sexual orientation and gender identity data in research and practice. American Journal of Public Health, 114(8), 777-781. https://doi.org/10.2105/AJPH.2024.307695
- Aguilera, D., Rinola, B., Tundealao, S., Klaff, R., Aung, M., Johnson-Campbell, M., Johnson-Wallace, D., Stephenson, R., Jolly, P. E., Tamí-Maury, I. (2024). Prevalence and predictors of cigarette smoking among people with HIV in Western Jamaica. AIDS Care, 36(10), 1499-1507. https://doi.org/10.1080/09540121.2024.2354224
- Tamí-Maury, I., Tundealao, S., Noé-Díaz, V., Garcia, E., Diaz, V., Meier, J., Dani, M., Vidaurre, T. (2024). Boosting self-efficacy and improving practices for smoking prevention and cessation among South American cancer care providers with a web-based algorithm. Addictive Sciences & Clinical Practice, 19(1), 36. https://doi.org/10.1186/s13722-024-00462-w
- De La Cruz-Vargas, J. A., Ramos, W., Chanduví, W., Correa-López, L. E., Guerrero, N., Loayza-Castro, J., Tami-Maury, I., Venegas, D. (2024). Proportion of cancer cases and deaths attributable to potentially modifiable risk factors in Peru. BMC Cancer, 24(1), 477. https://doi.org/10.1186/s12885-024-12219-4
- Smith, F. S., Lai, H., Tamí-Maury, I., Cornejo Gonzalez, A., Stuart, S., Denny, M. C., Ancer Leal, A., Sharrief, A., Maroufy, V., Savitz, S. I., Beauchamp, J. E. S. (2024). A cross-sectional survey of comprehension and satisfaction of Spanish-reading adults regarding RÁPIDO as a stroke awareness acronym. Journal of Neuroscience Nursing, 56(3), 69-74. https://doi.org/10.1097/JNN.0000000000000755
- Krenek, B., Tundealao, S., Beauchamp, J. E. S., Savitz, S. I., Tamí-Maury, I. (2024). Comparing stroke risk factors among sexual minority groups in Texas. International Journal of Behavioral Medicine. https://doi.org/10.1007/s12529-024-10267-5
- Choi, J., Cuccaro, P., Markham, C., Kim, S., Tamí-Maury, I. (2023). Human papillomavirus (HPV) vaccination in males: Associations of HPV-related knowledge and perceptions with HPV vaccination intention among Korean mothers of boys. Preventive Medicine Reports, 37, 102566. https://doi.org/10.1016/j.pmedr.2023.102566
- Klaff, R., Tundealao, S., Krenek, B., Tamí-Maury, I. (2023). Designing and pilot-testing SmokefreeSGM: A text-based smoking cessation intervention for sexual and gender minority groups. mHealth, 9, 23. https://doi.org/10.21037/mhealth-23-4
- Tamí-Maury, I., Tundealao, S., Díaz, V., Ochoa, E., Garcia, E., Rincon, J., Noé-Díaz, V., Castañeda, C., Acosta, J., Fernández, M., Vidaurre, T., Crowley, J. (2023). The STOP Program: A hybrid smoking prevention and cessation training for cancer care providers in Colombia and Peru. Journal of Cancer Education, 38(5), 1683-1689. https://doi.org/10.1007/s13187-023-02322-8
- Tamí-Maury, I., Tundealao, S., Guzman, J., Noé-Díaz, V., Markham, C., Vigil, K. (2023). Developing and pre-testing a digital decision-tree smartphone application for smoking prevention and cessation among HIV care providers. Digital Health, 9, 20552076231179029. https://doi.org/10.1177/20552076231179029
- Choi, J., Markham, C., Tamí-Maury, I., Kim, S., Cuccaro, P. (2023). Maternal perceptions of vaccinating boys against human papillomavirus (HPV) in Seoul, South Korea: A descriptive exploratory qualitative study. PLoS One, 18(3), e0282811. https://doi.org/10.1371/journal.pone.0282811
- Choi, J., Tamí-Maury, I., Cuccaro, P., Kim, S., Markham, C. (2023). Digital health interventions to improve adolescent HPV vaccination: A systematic review. Vaccines (Basel), 11(2), 249. https://doi.org/10.3390/vaccines11020249
-
Evan Kwiatkowski
- Yang C-H, Kwiatkowski E, Lee JJ, Lin R. REDOMA: Bayesian random-effects dose-optimization meta-analysis using spike-and-slab priors. Statistics in Medicine. 2024; 43(18): 3484-3502. doi: 10.1002/sim.10107
- Evan Kwiatkowski, Jiawen Zhu, Xiao Li, Herbert Pang, Grazyna Lieberman, Matthew A Psioda, Case weighted power priors for hybrid control analyses with time-to-event data, Biometrics, Volume 80, Issue 2, June 2024, ujae019, https://doi.org/10.1093/biomtc/ujae019
- Jibran Ahmed, Bettzy Stephen, Yali Yang, Evan Kwiatkowski, Chinenye Lynette Ejezie, Shubham Pant; Phase Ib/II Study of Lacnotuzumab in Combination with Spartalizumab in Patients with Advanced Malignancies. Journal of Immunotherapy and Precision Oncology 1 May 2024; 7 (2): 73–81. doi: https://doi.org/10.36401/JIPO-23-16
- Stephen B, Hajjar J, Sarda S, Duose DY, Conroy JM, Morrison C, Alshawa A, Xu M, Zarifa A, Patel SP, Yuan Y, Kwiatkowski E, Wang L, Rodon Ahnert J, Fu S, Meric-Bernstam F, Lowman GM, Looney T, Naing A. T-cell receptor beta variable gene polymorphism predicts immune-related adverse events during checkpoint blockade immunotherapy. J Immunother Cancer. 2023 Aug;11(8):e007236. doi: 10.1136/jitc-2023-007236. PMID: 37604642; PMCID: PMC10445351.
-
Ruosha Li
- Fwelo, P., Li, R., Heredia, N. I., Nyachoti, D., Adekunle, T. E., Adekunle, T. B., Bangolo, A., & Du, X. L. (2025). Disparities in thyroid cancer mortality across racial and ethnic groups: Assessing the impact of socioeconomic, clinicopathologic, and treatment variations. Annals of Surgical Oncology, 32(2), 1158-1175. https://doi.org/10.1245/s10434-024-16569-y
- Li, R., Wang, J., Zhang, C., Squires, J. E., Belle, S. H., Ning, J., Cai, J., & Squires, R. H. (2024). Improved mortality prediction for pediatric acute liver failure using dynamic prediction strategy. Journal of Pediatric Gastroenterology and Nutrition, 78(2), 320-327. https://doi.org/10.1002/jpn3.12094
- Stephens, S. B., Tsang, R., Li, R., Cazaban-Ganduglia, C., Agopian, A. J., & Morris, S. A. (2024). Congenital heart defects and concurrent diagnoses in influenza hospitalization in the Pediatric Health Information System study, 2004-2019. Pediatric Cardiology. Advance online publication. https://doi.org/10.1007/s00246-024-03613-7
- Li, F., Yano, Y., Étiévant, L., Daniel, C. R., Sharma, S. V., Brown, E. L., Li, R., Loftfield, E., Lan, Q., Sinha, R., Moshiree, B., Inoue-Choi, M., & Vogtmann, E. (2024). The time-dependent association between irritable bowel syndrome and all-cause and cause-specific mortality: A prospective cohort study within the UK Biobank. American Journal of Gastroenterology, 119(7), 1373-1382. https://doi.org/10.14309/ajg.0000000000002675
- Li, W., Li, R., Feng, Z., & Ning, J.; Alzheimer’s Disease Neuroimaging Initiative. (2024). Dynamic and concordance-assisted learning for risk stratification with application to Alzheimer's disease. Advance online publication. https://doi.org/10.1093/biostatistics/kxae036
- Peña, M. T., Lindsay, J. A., Li, R., Deshmukh, A. A., Swint, J. M., & Morgan, R. O. (2024). Telemental health use is associated with lower health care spending among Medicare beneficiaries with major depression. Medical Care, 62(3), 132-139. https://doi.org/10.1097/MLR.0000000000001952
- Liu, X., Ning, J., He, X., Tilley, B. C., & Li, R. (2023). Semiparametric regression modeling of the global percentile outcome. Journal of Statistical Planning and Inference, 222, 149-159. https://doi.org/10.1016/j.jspi.2022.06.009
- Zhang, C., Ning, J., Cai, J., Squires, J. E., Belle, S. H., & Li, R. (2024). Dynamic risk score modeling for multiple longitudinal risk factors and survival. Computational Statistics & Data Analysis, 189, 107837. https://doi.org/10.1016/j.csda.2023.107837
- Backley, S., Bergh, E., Garnett, J., Li, R., Maroufy, V., Jain, R., Fletcher, S., Tsao, K., Austin, M., Johnson, A., & Papanna, R. (2024). Fetal cardiovascular changes during open and fetoscopic in-utero spina bifida closure. Ultrasound in Obstetrics & Gynecology. Advance online publication. https://doi.org/10.1002/uog.27579
- Guo, X., Li, R., Zhou, J., & He, X. (2023). A robust quantitative risk screening for subgroup pursuit in clinical trials. Econometrics and Statistics. Manuscript in preparation.
- Li, F., Ramirez, Y., Yano, Y., Daniel, C. R., Sharma, S. V., Brown, E. L., Li, R., Moshiree, B., Loftfield, E., Lan, Q., Sinha, R., Inoue-Choi, M., & Vogtmann, E. (2023). The association between inflammatory bowel disease and all-cause and cause-specific mortality in the UK Biobank. Annals of Epidemiology, 88, 15-22. https://doi.org/10.1016/j.annepidem.2023.10.008
-
Jose Miguel Yamal
2025:
Rajan, S. S., Yamal, J. M., Wang, M., Saver, J. L., Jacob, A. P., Gonzales, N. R., Ifejika, N., Parker, S. A., Ganey, C., Gonzalez, M. O., Lairson, D. R., Bratina, P. L., Jones, W. J., Mackey, J. S., Lerario, M. P., Navi, B. B., Alexandrov, A. W., Alexandrov, A., Nour, M., Spokoyny, I., Bowry, R., Czap, A. L., & Grotta, J. C. (2025). A prospective multicenter analysis of mobile stroke unit cost-effectiveness. Annals of Neurology, 97(2), 209-221. https://doi.org/10.1002/ana.27105
2024:
- Yamal J-M, Mofleh D, Chuang R-J, Wang M, Johnson K, Garcia-Quintana A, Titiloye T, Nelson S, Sharma S. Training Protocol and Calibration of the International Caries Detection and Assessment System in a School-Based Clinical Trial of Elementary School-Age Children. J of Public Health Dentistry, 08 December 2024 https://doi.org/10.1111/jphd.12648.
- Tiruneh YM, Cuccaro PM, Elliott KS, Xie J, Martinez J, Owens M, Alvarado C, Yamal J-M§. Vaccine Uptake and Intentions: Insights from a Texas Survey on Factors Influencing COVID-19 Vaccination Decisions. Vaccines, in press.
- Jannace KC*, Pompeii L, Gimeno Ruiz de Porras D, Perkison WB, Yamal J-M, Trone DW, Rull RP. Risk of Traumatic Brain Injury In Deployment and Nondeployment Settings Among Members of the Millennium Cohort Study. Journal of Head Trauma Rehabilitation, in press.
- Cuccaro PM, Choi J, Tiruneh YM, Martinez J, Xie J, Crum M, Owens M, Yamal J-M§. Parental factors associated with COVID-19 vaccine uptake for children over 5 years of age in Texas. Vaccines. 2024 May 11;12(5):526.
- Navi BB, Bach I, Czap AL, Wang M, Yamal J-M, Jacob AP, Parker SA, Rajan SS, Mir S, Sherman C, Willey JZ, Saver JL, Gonzalez MO, Singh N, Jones WJ, Ornelas D, Gonzales NR, Alexandrov AW, Alexandrov AV, Nour M, Spokoyny I, Mackey J, Collins SQ, Silnes K, Fink ME, English J, Barazangi N, Bratina PL, Volpi J, Rao CPV, Grivvin L, Persse D, Grotta JC. Strokes Averted by Intravenous Thrombolysis: A Secondary Analysis of a Prospective, Multicenter, Controlled Trial of Mobile Stroke Units. Annals of Neurology. 2024 Feb;95(2):347-61.
-
Samiran Ghosh
- Majumder, P., Mukhopadhyay, S., Wang, B., & Ghosh, S. (2025). Sample size determinations in four-level longitudinal cluster randomized trials with random slope. Statistical Methods in Medical Research. (In press)
- Belzer, M. E., MacDonell, K., Cain, D., Ghosh, S., Zhao, R., McAvoy-Banerjea, J., Gurung, S., & Naar, S. (2024). An adaptive antiretroviral therapy adherence intervention for youth with HIV through text message and cell phone support with and without incentives: A sequential multiple assignment randomized trial (SMART). AIDS and Behavior. Advance online publication. https://doi.org/10.1007/s10461-024-04558-x
- Ghosh, S., Banerjee, M., & Chattopadhyay, A. K. (2024). Effect of vaccine dose intervals: Considering immunity levels, vaccine efficacy, and strain variants for disease control strategy. PLOS ONE, 19(9), Article e0310152. https://doi.org/10.1371/journal.pone.0310152
- Zheng, S., Mansolf, M., McGrath, M., Churchill, M. L., Bekelman, T. A., Brennan, P. A., Margolis, A. E., Nozadi, S. S., Bastain, T. M., Elliott, A. J., LeWinn, K. Z., Hofheimer, J. A., Leve, L. D., Rennie, B., Zimmerman, E., Marable, C. A., McEvoy, C. T., Liu, C., Sullivan, A., ... Bishop, S. (2023). Measurement bias in caregiver-report of early childhood behavior problems across demographic factors in an ECHO-wide diverse sample. JCPP Advances, 4(1), Article e12198. https://doi.org/10.1002/jcv2.12198
- Paul, E., Chakraborty, B., Sikorskii, A., & Ghosh, S. (2024). A framework for testing non-inferiority in a three-arm, sequential, multiple assignment randomized trial. Statistical Methods in Medical Research, 33(4), 611-633. https://doi.org/10.1177/09622802241232124
- Cunningham, P. B., Naar, S., Roberts, J. R., Powell, J., Ledgerwood, D. M., Randall, J., Lozano, B. E., Halliday, C. A., Madisetti, M., & Ghosh, S. (2024). Study protocol for clinical trial of the FIT Families multicomponent obesity intervention for African American adolescents and their caregivers: Next step from the ORBIT initiative. BMJ Open, 14(2), Article e074552. https://doi.org/10.1136/bmjopen-2023-074552
- Kvale, E., Phillips, F., Ghosh, S., Lea, J., Hoppenot, C., Costales, A., Sunde, J., Badr, H., Nwogu-Onyemkpa, E., Saleem, N., Ward, R., & Balasubramanian, B. (2024). Survivorship care for women living with ovarian cancer: Protocol for a randomized controlled trial. JMIR Research Protocols, 13, Article e48069. https://doi.org/10.2196/48069
- Saade, M., Ghosh, S., Banerjee, M., & Volpert, V. (2024). Delay epidemic models determined by latency, infection, and immunity duration. Mathematical Biosciences, 370, Article 109155. https://doi.org/10.1016/j.mbs.2024.109155
- Bhowmick, R. S., Sarkar, A., Ghosh, S., Gope, S., & Chakraborty, R. (2023). Postoperative pulmonary complication as an emerging complication in major head and neck cancer surgery: A retrospective study. National Journal of Maxillofacial Surgery, 14(3), 471-476. https://doi.org/10.4103/njms.njms_399_21
- Schieber, E., Deveaux, L., Cotrell, L., Li, X., Lemon, S. C., Ash, A. S., MacDonell, K., Ghosh, S., Poitier, M., Rolle, G., Naar, S., & Wang, B. (2024). Maintaining program fidelity in a changing world: National implementation of a school-based HIV prevention program. Prevention Science, 25(3), 436-447. https://doi.org/10.1007/s11121-023-01614-1
- Ghosh, S., Ogueda-Oliva, A., Ghosh, A., Banerjee, M., & Seshaiyer, P. (2023). Understanding the implications of under-reporting, vaccine efficiency and social behavior on the post-pandemic spread using physics informed neural networks: A case study of China. PLOS ONE, 18(11), Article e0290368. https://doi.org/10.1371/journal.pone.0290368
- Cunningham, P. B., Gilmore, J., Naar, S., Preston, S. D., Eubanks, C. F., Hubig, N. C., McClendon, J., Ghosh, S., & Ryan-Pettes, S. (2023). Opening the black box of family-based treatments: An artificial intelligence framework to examine therapeutic alliance and therapist empathy. Clinical Child and Family Psychology Review, 26(4), 975-993. https://doi.org/10.1007/s10567-023-00451-6
- Saade, M., Ghosh, S., Banerjee, M., & Volpert, V. (2023). An epidemic model with time delays determined by the infectivity and disease durations. Mathematical Biosciences and Engineering, 20(7), 12864-12888. https://doi.org/10.3934/mbe.2023574
- Buenconsejo, J., Liao, R., Lin, J., Singh, P., Cooner, F., Ghosh, S., Gamalo, M., Russek-Cohen, E., & Zariffa, N. (2023). Platform trials to evaluate the benefit-risk of COVID-19 therapeutics: Successes, learnings, and recommendations for future pandemics. Contemporary Clinical Trials, 132, Article 107292. https://doi.org/10.1016/j.cct.2023.107292
- Cannoy, C. N., Bauer, S. J., Prakash, K., Excell, S., Ghosh, S., Lundahl, L. H., & Ledgerwood, D. M. (2023). Response to health warnings on cigarette packs as a predictor of future smoking among current tobacco smokers. Addictive Behaviors, 144, Article 107717. https://doi.org/10.1016/j.addbeh.2023.107717
- Sarkar, A., Banerjee, L., & Ghosh, S. (2023). Efficacy of Delta Plate in condylar fracture: A case series with review. Journal of Dentistry, Shiraz, 24(1), 71-75. https://doi.org/10.30476/DENTJODS.2022.94746.1806
- Ghosh, S., Volpert, V., & Banerjee, M. (2023). An age-dependent immuno-epidemiological model with distributed recovery and death rates. Journal of Mathematical Biology, 86(2), Article 21. https://doi.org/10.1007/s00285-022-01855-8
-
Dejian Li
- Agarwal, N., Papanna, R., Sibai, B. M., Garcia, A., Lai, D., Soto Torres, E. E., ... & Hernandez-Andrade, E. (2024). Evaluation of fetal growth and birth weight in pregnancies with placenta previa with and without placenta accreta spectrum. Journal of Perinatal Medicine. Advance online publication.
- Agarwal, N., Hernandez-Andrade, E., Sibai, B. M., Amro, F. H., Coselli, J. O., Bartal, M. F., ... & Papanna, R. (2024). Quantifying placenta accreta spectrum severity and its associated blood loss: A novel transvaginal ultrasound scoring system. American Journal of Obstetrics & Gynecology MFM, 6(10), 101451.
- Agarwal, N. D., Bartal, M. F., Coselli, J., Torres, E. E. S., Andrade, E. H., Lai, D., ... & Papanna, R. (2024). Transvaginal/Transvesical Ultrasound (TVUS) to diagnose placenta accreta spectrum (PAS), its severity & to predict blood loss. American Journal of Obstetrics & Gynecology, 230(1), S444-S445.
- Backley, S., Agarwal, N. D., Weitzel, L., Lai, D., Bergh, E. P., Johnson, A., ... & Papanna, R. (2024). Does fetoscopic laser ablation (FLA) improve perinatal outcomes in Types II and III Vasa Previa? American Journal of Obstetrics & Gynecology, 230(1), S214-S215.
- Gould, K. L., Johnson, N. P., Roby, A. E., Bui, L., Kitkungvan, D., Patel, M. B., ... & Narula, J. (2024). Coronary flow capacity and survival prediction after revascularization: Physiological basis and clinical implications. European Heart Journal, 45(3), 181-194.
- Gould, K. J., Nguyen, T., Kirkeeide, R., Roby, A. E., Bui, L., Kitkungvan, D., Patel, M. B., Madjid, M., Haynie, M., Lai, D., Li, R., Narula, J., & Johnson, N. P. (2023). Subendocardial and transmural myocardial ischemia clinical characteristics, prevalence, and outcomes with and without revascularization. JACC: Cardiovascular Imaging, 16, 78-94.
- Hoffman, J. R., Park, H. J., Bheri, S., Platt, M. O., Hare, J. M., Kaushal, S., Bettencourt, J. L., Lai, D., Slesnick, T. C., Mahle, W. T., & Davis, M. E. (2023). Statistical modeling of extracellular vesicle cargo to predict clinical trial outcomes for hypoplastic left heart syndrome. https://doi.org/10.1016/j.isci.2023.107980
- Li, C., Lai, D., Jiang, X., & Zhang, K. (2024). FERI: A multitask-based fairness achieving algorithm with applications to fair organ transplantation. AMIA Summits on Translational Science Proceedings, 2024, 593.
- Perkison, W. B., Shegog, R., Lai, D., Upadhyay, S., Yalavarthy, G., Guerrero-Luera, R., ... & Nabeel, I. (2024). Development of system-based digital decision support (“Pocket Ark”) for post-flood enhanced response coordination and worker safety: An intervention mapping approach. Frontiers in Environmental Health, 3, 1368077.
- Xia, L., Chen, B., & Lai, D. (2024). Group sequential tests by boundary crossing via drift fractional Brownian motion: Bayesian estimation. Communications in Statistics-Simulation and Computation, 1-9.
- Xia, L., Chen, B., & Lai, D. (2024). Sequential monitoring clinical trial by conditional power under drift fractional Brownian motion. Communications in Statistics-Simulation and Computation, 53(10), 4676-4684.
- Ye, J., & Lai, D. (2024). Covariate adjusted nonparametric methods under propensity analysis. Frontiers in Applied Mathematics and Statistics, 10, 1357816.
- Zheng, W., Zhu, H., Lance Gould, K., & Lai, D. (2024). Comparing heart PET scans: An adjustment of Kolmogorov-Smirnov test under spatial autocorrelation. Journal of Applied Statistics, 1-17.
- Zheng, W. J., Lai, D., & Gould, K. L. (2023). A simulation study of a class of nonparametric test statistics: A close look at the Kolmogorov-Smirnov test. Communications in Statistics-Simulation and Computation, 52, 1133-1148. https://doi.org/10.1080/03610918.2021.1874987
- Zhu, H., Yu, J., Wang, Q., Lai, D., Wang, L., & Zhong, S. (2024). Adaptive seamless phase II/III design with sequential estimation-adjusted urn model. Communications in Statistics-Simulation and Computation, 1-11.
- Zhu, H. J., Yu, J., & Lai, D. (2023). Seamless clinical trials with doubly adaptive biased coin designs. New England Journal of Statistics in Data Science, 1, 314-322.
-
Luis N. Nevelo
- Sherer, M., Juengst, S., Sander, A. M., Leon-Novelo, L., Liu, X., Bogaards, J., Chua, W., & Tran, K. (2025). Mood tracker: A randomized controlled trial of a self-monitoring intervention for emotional distress after traumatic brain injury. Journal of Head Trauma Rehabilitation, 40(1), E13-E22. https://doi.org/10.1097/HTR.0000000000000945
-
Vahed Maroufy
- Yoshida, A., Li, Y., Maroufy, V., Kuwana, M., Sazliyana Shaharir, S., Makol, A., Sen, P., Lilleker, J. B., Agarwal, V., Kadam, E., Akawatcharangura Goo, P., Day, J., Milchert, M., Chen, Y. M., Dey, D., Velikova, T., Saha, S., Edgar Gracia-Ramos, A., Parodis, I., ... Gupta, L.; COVAD Study Group. (2024). Impaired health-related quality of life in idiopathic inflammatory myopathies: A cross-sectional analysis from the COVAD-2 e-survey. Rheumatology Advances in Practice, 8(2), rkae028. https://doi.org/10.1093/rap/rkae028
- Williams, G. W., Mubashir, T., Balogh, J., Rezapour, M., Hu, J., Dominique, B., Gautam, N. K., Lai, H., Ahmad, H. S., Li, X., Huang, Y., Zhang, G. Q., & Maroufy, V. (2023). Recent COVID-19 infection is associated with increased mortality in the ambulatory surgery population. Journal of Clinical Anesthesia, 89, 111182. https://doi.org/10.1016/j.jclinane.2023.111182
- Boren, S. B., Savitz, S. I., Gonzales, N., Hasan, K., Becerril-Gaitan, A., Maroufy, V., Li, Y., Grotta, J., Steven, E. A., Chen, C. J., Sitton, C. W., Aronowski, J., & Haque, M. E. (2024). Longitudinal morphometric changes in the corticospinal tract shape after hemorrhagic stroke. Translational Stroke Research, 15(5), 893–901. https://doi.org/10.1007/s12975-023-01168-y
- Testa, A., Jackson, D. B., Gutierrez, C., Fahmy, C., Maroufy, V., Samper-Ternent, R., & Neumann, A. C. (2024). History of incarceration and dental care use among older adults in the United States. American Journal of Preventive Medicine, 67(5), 705–712. https://doi.org/10.1016/j.amepre.2024.06.023
- Testa, A., Mungia, R., Neumann, A. C., Samper-Ternent, R., Mijares, L., Maroufy, V., Ganson, K. T., Nagata, J. M., & Jackson, D. B. (2024). Prior incarceration length and edentulism among formerly incarcerated older adults in the United States. Journal of the American Dental Association, 155(11), 935–944.e3. https://doi.org/10.1016/j.adaj.2024.08.016
- Tsai, J., Lai, H., & Maroufy, V. (2024). Longitudinal change and association between four types of social support and mental health among low-income U.S. veterans. International Journal of Social Psychiatry. Advance online publication. https://doi.org/10.1177/00207640241299321
- Tsai, J., Li, Y., & Maroufy, V. (2024). Prevalence of four types of social support and their relation to mental health among low-income U.S. veterans: Implications for community health. Community Mental Health Journal, 60(8), 1652–1662. https://doi.org/10.1007/s10597-024-01318-y
- Smith, F. S., Lai, H., Tamí-Maury, I., Cornejo Gonzalez, A., Stuart, S., Denny, M. C., Ancer Leal, A., Sharrief, A., Maroufy, V., Savitz, S. I., & Beauchamp, J. E. S. (2024). A cross-sectional survey of comprehension and satisfaction of Spanish-reading adults regarding RÁPIDO as a stroke awareness acronym. Journal of Neuroscience Nursing, 56(3), 69–74. https://doi.org/10.1097/JNN.0000000000000755
-
Shreela Sharma
- Volpp, K. G., Berkowitz, S. A., Sharma, S. V., Anderson, C. A. M., Brewer, L. C., Elkind, M. S. V., Gardner, C. D., Gervis, J. E., Harrington, R. A., Herrero, M., Lichtenstein, A. H., McClellan, M., Muse, J., Roberto, C. A., & Zachariah, J. P. V. (2023). Food is medicine: A presidential advisory from the American Heart Association. Circulation, 148(18), 1417–1439. https://doi.org/10.1161/CIR.0000000000001182
- Li, F., Ramirez, Y., Yano, Y., Daniel, C. R., Sharma, S. V., Brown, E. L., Li, R., Moshiree, B., Loftfield, E., Lan, Q., Sinha, R., Inoue-Choi, M., & Vogtmann, E. (2023). The association between inflammatory bowel disease and all-cause and cause-specific mortality in the UK Biobank. Annals of Epidemiology, 88, 15–22. https://doi.org/10.1016/j.annepidem.2023.10.008
- Li, F., Yano, Y., Étiévant, L., Daniel, C. R., Sharma, S. V., Brown, E. L., Li, R., Loftfield, E., Lan, Q., Sinha, R., Moshiree, B., Inoue-Choi, M., & Vogtmann, E. (2024). The time-dependent association between irritable bowel syndrome and all-cause and cause-specific mortality: A prospective cohort study within the UK Biobank. American Journal of Gastroenterology, 119(7), 1373–1382 https://doi.org/10.14309/ajg.0000000000002675
- Metoyer, B. N., Chuang, R. J., Lee, M., Markham, C., Brown, E., Almohamad, M., & Sharma, S. V. (2023). SNAP participation moderates fruit and vegetable intake among minority families with low income. Journal of Nutrition Education and Behavior, 55(11), 774–785. https://doi.org/10.1016/j.jneb.2023.08.005
- Davis, J. N., Nikah, K., Landry, M. J., Vandyousefi, S., Ghaddar, R., Jeans, M., Cooper, M. H., Martin, B., Waugh, L., Sharma, S. V., & van den Berg, A. E. (2023). Effects of a school-based garden program on academic performance: A cluster randomized controlled trial. Journal of the Academy of Nutrition and Dietetics, 123(4), 637–642. https://doi.org/10.1016/j.jand.2022.08.125
- McWhorter, J. W., Aiyer, J. N., Ranjit, N., Toups, J., Liew, E., John, J. C., & Sharma, S. V. (2023). Perspectives of health care staff on predictors of success in a food prescription program: A qualitative study. Preventing Chronic Disease, 20, E02. https://doi.org/10.5888/pcd20.220178
- Naylor Metoyer, B., Chuang, R. J., Lee, M., Markham, C., Brown, E. L., Almohamad, M., Dave, J. M., & Sharma, S. V. (2023). Fruit and vegetable intake and home nutrition environment among low-income minority households with elementary-aged children. Nutrients, 15(8), 1819. https://doi.org/10.3390/nu15081819
- Ranjit, N., Aiyer, J. N., Toups, J. D., Liew, E., Way, K., Brown, H. S., McWhorter, J. W., & Sharma, S. V. (2023). Clinical outcomes of a large-scale, partnership-based regional food prescription program: Results of a quasi-experimental study. BMC Research Notes, 16(1), 13. https://doi.org/10.1186/s13104-023-06280-8
- John, J. C., Gonzalez, J., Chan, S. G., McPherson, H., Aiyer, J. N., Galvan, E., Browning, N., & Sharma, S. V. (2023). A coalition-driven examination of organization capacity to address food insecurity in Greater Houston: A qualitative research study. Frontiers in Public Health, 11, 1167100. https://doi.org/10.3389/fpubh.2023.1167100
- Haushalter, K., Burgermaster, M., Hudson, E., Landry, M. J., Sharma, S. V., & Davis, J. N. (2024). An increase in food insecurity correlated with an increase in plasma triglycerides among Latinx children. Journal of Nutrition, 154(2), 565–573. https://doi.org/10.1016/j.tjnut.2023.12.024
- Craig, D. W., Walker, T. J., Sharma, S. V., Cuccaro, P., Heredia, N. I., Pavlovic, A., DeFina, L. F., Kohl, H. W. 3rd, & Fernandez, M. E. (2024). Examining associations between school-level determinants and the implementation of physical activity opportunities. Translational Behavioral Medicine, 14(2), 89–97. https://doi.org/10.1093/tbm/ibad055
- Craig, D. W., Walker, T. J., Cuccaro, P., Sharma, S. V., Heredia, N. I., Robertson, M. C., & Fernandez, M. E. (2024). Using the R = MC(2) heuristic to understand barriers to and facilitators of implementing school-based physical activity opportunities: A qualitative study. BMC Public Health, 24(1), 207. https://doi.org/10.1186/s12889-024-17744-2
-
Lara Simpson
- Cholesterol Treatment Trialists’ (CTT) Collaboration. (2024). Effects of statin therapy on diagnoses of new-onset diabetes and worsening glycaemia in large-scale randomised blinded statin trials: An individual participant data meta-analysis. The Lancet Diabetes & Endocrinology, 12(5), 306–319. https://doi.org/10.1016/S2213-8587(24)00040-8
- Yamal, J. M., Martinez, J., Osani, M. C., Du, X. L., Simpson, L. M., & Davis, B. R. (2023). Mortality and morbidity among individuals with hypertension receiving a diuretic, ACE inhibitor, or calcium channel blocker: A secondary analysis of a randomized clinical trial. JAMA Network Open, 6(12), e2344998. https://doi.org/10.1001/jamanetworkopen.2023.44998
- Wu, R., Williams, C., Zhou, J., Schlackow, I., Emberson, J., Reith, C., Keech, A., Robson, J., Armitage, J., Gray, A., Simes, J., Baigent, C., Mihaylova, B., & CTT Collaboration. (2024). Long-term cardiovascular risks and the impact of statin treatment on socioeconomic inequalities: A microsimulation model. British Journal of General Practice, 74(740), e189–e198. https://doi.org/10.3399/BJGP.2023.0198
- Du, X. L., Martinez, J., Yamal, J. M., Simpson, L. M., & Davis, B. R. (2023). The 18-year risk of cancer, angioedema, insomnia, depression, and erectile dysfunction in association with antihypertensive drugs: Post-trial analyses from ALLHAT-Medicare linked data. Frontiers in Cardiovascular Medicine, 10, 1272385. https://doi.org/10.3389/fcvm.2023.1272385
-
Michael D. Swartz
- Fowler, S. P., Gimeno Ruiz de Porras, D., Swartz, M. D., Stigler Granados, P., Heilbrun, L. P., & Palmer, R. F. (2023). Daily early-life exposures to diet soda and aspartame are associated with autism in males: A case-control study. Nutrients, 15(17), 3772. https://doi.org/10.3390/nu15173772
- Sandoval, M. N., McClellan, S. P., Pont, S. J., Ross, J. A., Swartz, M. D., Silberman, M. A., & Boerwinkle, E. (2024). Prozone masks elevated SARS-CoV-2 antibody level measurements. PLoS One, 19(3), e0301232. https://doi.org/10.1371/journal.pone.0301232
- Swartz, M. D., DeSantis, S. M., Yaseen, A., Brito, F. A., Valerio-Shewmaker, M. A., Messiah, S. E., Leon-Novelo, L. G., Kohl, H. W., Pinzon-Gomez, C. L., Hao, T., Zhang, S., Talebi, Y., Yoo, J., Ross, J. R., Gonzalez, M. O., Wu, L., Kelder, S. H., Silberman, M., Tuzo, S., Pont, S. J., Shuford, J. A., Lakey, D., & Boerwinkle, E. (2023). Antibody duration after infection from SARS-CoV-2 in the Texas Coronavirus Antibody Response Survey. The Journal of Infectious Diseases, 227(2), 193–201. https://doi.org/10.1093/infdis/jiac167
- Song, J., Swartz, M. D., & Basen-Engquist, K. (2023). acc: An R package to process, visualize, and analyze accelerometer data. Software Impacts, 18, 100577. https://doi.org/10.1016/j.simpa.2023.100577
- Ish, J., Symanski, E., Gimeno Ruiz de Porras, D., Casas, M., Delclos, G. L., Guxens, M., Ibarluzea, J. M., Iñiguez, C., Lertxundi, A., Rebagliato, M., Swartz, M. D., & Whitworth, K. W. (2023). Correction: Maternal occupational exposure to chemicals and child cognitive function. Pediatric Research, 93(1), 278-279. https://doi.org/10.1038/s41390-022-02300-8
- Kendzor, D. E., Businelle, M. S., Frank-Pearce, S. G., Waring, J. J. C., Chen, S., Hébert, E. T., Swartz, M. D., Alexander, A. C., Sifat, M. S., Boozary, L. K., & Wetter, D. W. (2024). Financial incentives for smoking cessation among socioeconomically disadvantaged adults: A randomized clinical trial. JAMA Network Open, 7(7), e2418821. https://doi.org/10.1001/jamanetworkopen.2024.18821
- Nyitray, A. G., Nitkowski, J., McAuliffe, T. L., Brzezinski, B., Swartz, M. D., Fernandez, M. E., Deshmukh, A. A., Ridolfi, T. J., Lundeen, S. J., Cockerham, L., Wenten, D., Petroll, A., Hilgeman, B., Smith, J. S., Chiao, E. Y., Giuliano, A. R., & Schick, V. (2023). Home-based self-sampling vs clinician sampling for anal precancer screening: The Prevent Anal Cancer Self-Swab Study. International Journal of Cancer, 153(4), 843-853. https://doi.org/10.1002/ijc.34553
- Zhang, S., Chou, L. N., Swartz, M. D., Mehta, H. B., Goodwin, J. S., Kuo, Y. F., Giordano, S. H., Tucker, C. A., Basen-Engquist, K. M., Lyons, E. J., Downer, B., Peterson, S. K., Cao, T., & Swartz, M. C. (2024). Association of cancer diagnosis with disability status among older survivors of colorectal cancer: A population-based retrospective cohort study. Frontiers in Oncology, 14, 1283252. https://doi.org/10.3389/fonc.2024.1283252
- DeSantis, S. M., Yaseen, A., Hao, T., León-Novelo, L., Talebi, Y., Valerio-Shewmaker, M. A., Pinzon Gomez, C. L., Messiah, S. E., Kohl, H. W., Kelder, S. H., Ross, J. A., Padilla, L. N., Silberman, M., Wylie, S., Lakey, D., Shuford, J. A., Pont, S. J., Boerwinkle, E., & Swartz, M. D. (2023). RE: Incidence of SARS-CoV-2 breakthrough infections after vaccination in adults: A population-based survey through 1 March 2023. Open Forum Infectious Diseases, 10(12), ofad564. https://doi.org/10.1093/ofid/ofad564
- Chen, W. J., Rector-Houze, A. M., Guxens, M., Iñiguez, C., Swartz, M. D., Symanski, E., Ibarluzea, J., Valentin, A., Lertxundi, A., González-Safont, L., Sunyer, J., & Whitworth, K. W. (2024). Susceptible windows of prenatal and postnatal fine particulate matter exposures and attention-deficit hyperactivity disorder symptoms in early childhood. Science of the Total Environment, 912, 168806. https://doi.org/10.1016/j.scitotenv.2023.168806
Contact us
Interested in collaborating?
The University of Texas Health Science Center
School of Public Health
1200 Pressler St., 8th and 9th Floor
Houston, Texas 77030
Phone: 713-500-9529
Email: [email protected]